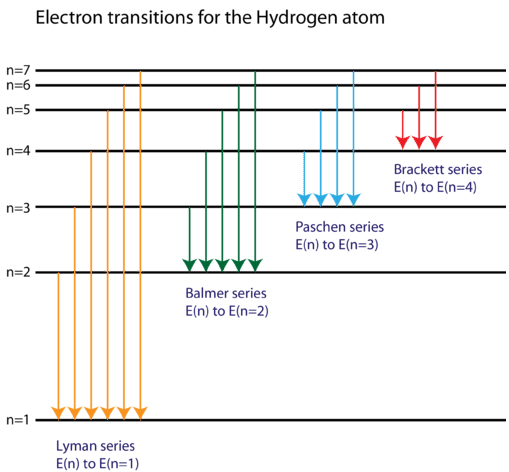 
In the 1880s, Williamina Fleming devised a system to classify stars based on the strength of hydrogen absorption lines. You will see, as we tell you the history, that it’s an instance where tradition won out over common sense. Recently, astronomers have added three additional classes for even cooler objects-L, T, and Y.Īt this point, you may be looking at these letters with wonder and asking yourself why astronomers didn’t call the spectral types A, B, C, and so on. From hottest to coldest, these seven spectral classes are designated O, B, A, F, G, K, and M. There are seven standard spectral classes. Because a star’s temperature determines which absorption lines are present in its spectrum, these spectral classes are a measure of its surface temperature. Classification of Stellar SpectraĪstronomers use the patterns of lines observed in stellar spectra to sort stars into a spectral class. Similarly, every other chemical element, in each of its possible stages of ionization, has a characteristic temperature at which it is most effective in producing absorption lines in any particular part of the spectrum. They can then absorb additional photons, rise to still-higher levels of excitation, and produce a dark absorption line. At this temperature, an appreciable number of hydrogen atoms are excited to the second energy level. Calculations show that the optimum temperature for producing visible hydrogen lines is about 10,000 K. The hydrogen lines in the visible part of the spectrum (called Balmer lines) are strongest in stars with intermediate temperatures-not too hot and not too cold. What this means is that if you observe the spectrum of a very hot or very cool star with a typical telescope on the surface of Earth, the most common element in that star, hydrogen, will show very weak spectral lines or none at all. Photons with enough energy to do this lie in the ultraviolet part of the electromagnetic spectrum, and there are very few ultraviolet photons in the radiation from a cool star. However, practically all of the hydrogen atoms are in the lowest energy state (unexcited) in these stars and thus can absorb only those photons able to lift an electron from that first energy level to a higher level. In the atmospheres of the coolest stars, hydrogen atoms have their electrons attached and can switch energy levels to produce lines. (Recall from the Formation of Spectral Lines section, the lines are the result of electrons in orbit around a nucleus changing energy levels.) Because the electron and the proton are separated, ionized hydrogen cannot produce absorption lines. In the atmospheres of the hottest stars, hydrogen atoms are completely ionized. However, lines of hydrogen are not seen in the spectra of the hottest and the coolest stars. Hydrogen, for example, is by far the most abundant element in most stars. 
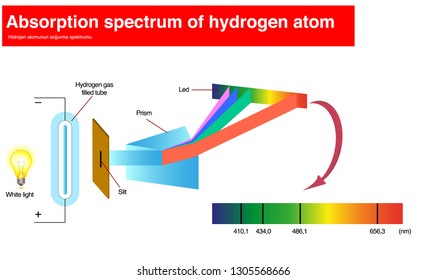
Most stars have nearly the same composition as the Sun, with only a few exceptions. The primary reason that stellar spectra look different is because the stars have different temperatures. Since the dark lines are produced by the chemical elements present in the stars, astronomers first thought that the spectra differ from one another because stars are not all made of the same chemical elements. When the spectra of different stars were first observed, astronomers found that they were not all identical. William and Margaret Huggins were the first to identify the lines in the spectrum of a star other than the Sun they also took the first spectrogram, or photograph of a stellar spectrum. Today, spectroscopic analysis is one of the cornerstones of astronomical research.įigure 17.4 William Huggins (1824–1910) and Margaret Huggins (1848–1915). Since then, astronomers have worked hard to perfect experimental techniques for obtaining and measuring spectra, and they have developed a theoretical understanding of what can be learned from spectra. In the 1860s, English astronomers Sir William Huggins and Lady Margaret Huggins ( Figure 17.4) succeeded in identifying some of the lines in stellar spectra as those of known elements on Earth, showing that the same chemical elements found in the Sun and planets exist in the stars. In 1814, the German physicist Joseph Fraunhofer observed that the spectrum of the Sun shows dark lines crossing a continuous band of colors. Another way is to use a spectrograph to spread out the light into a spectrum (see the Radiation and Spectra and the Astronomical Instruments chapters). Measuring colors is only one way of analyzing starlight. Explain the difference between a star and a brown dwarf.Describe how astronomers use spectral classes to characterize stars.By the end of this section, you will be able to:
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
